Flame Test Lab
Skylar Haynes
8/15/14: We started this lab on friday the 12th and ended it on monday the 15th.
The purpose of this lab was to see how different chemicals create different color flames when set on fire. First each day of this lab right when we walked in the door we put on equipment such as goggles and aprons. Then we would sit down with our partners at a table with a tray. Then between the 2 of us we decided who was gonna be the mechanical handler and the runner. The first day I was the runner and my partner was the mechanical handler then the 2nd day of the lab we switched roles. Then the runner of the group we took the watch glasses and went up to our teacher to get the chemical. The the mechanical handler would add 5 drops of methanal. After that we used the stir stick to stir the liquid and the powder. After that we lit a match and carefully set the outside liquid on fire. Then we blew out the match away from the watch glass. We then watched the flame burn as the colors changed. Then we would record the data on our flame lab table. We did this procedure for each and every 7 different chemicals. Except in the middle of each chemical we would clean the stir stick and the watch glass.
PRE-LAB
1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
RED LIGHT
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
VIOLET LIGHT
3. What is the definition of a salt in chemistry?
VIOLET LIGHT
4. What is fire?
RED LIGHT
5. What are the chemicals involved in lighting a match?
THEY COULD BE HEATED BY AN OUTSIDE SOURCE OR ABSORBED BY PROTONS
6. What is a chemical reaction?
THEY ARE IN HIGH ENERGY STATE
7. What natural cycles does wildfire have an impact on?
HIGHT ENOUGH ENERGY THEY EMIT PROTONS SOME ELEMENTS REQUIRED HIGHER ENERGY
8. List at least three impacts of wildfire in San Diego.
WATER THAT IS WASTED, ANIMALS DYING, NATURE IS BURNING
8/15/14: We started this lab on friday the 12th and ended it on monday the 15th.
The purpose of this lab was to see how different chemicals create different color flames when set on fire. First each day of this lab right when we walked in the door we put on equipment such as goggles and aprons. Then we would sit down with our partners at a table with a tray. Then between the 2 of us we decided who was gonna be the mechanical handler and the runner. The first day I was the runner and my partner was the mechanical handler then the 2nd day of the lab we switched roles. Then the runner of the group we took the watch glasses and went up to our teacher to get the chemical. The the mechanical handler would add 5 drops of methanal. After that we used the stir stick to stir the liquid and the powder. After that we lit a match and carefully set the outside liquid on fire. Then we blew out the match away from the watch glass. We then watched the flame burn as the colors changed. Then we would record the data on our flame lab table. We did this procedure for each and every 7 different chemicals. Except in the middle of each chemical we would clean the stir stick and the watch glass.
PRE-LAB
1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
RED LIGHT
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
VIOLET LIGHT
3. What is the definition of a salt in chemistry?
VIOLET LIGHT
4. What is fire?
RED LIGHT
5. What are the chemicals involved in lighting a match?
THEY COULD BE HEATED BY AN OUTSIDE SOURCE OR ABSORBED BY PROTONS
6. What is a chemical reaction?
THEY ARE IN HIGH ENERGY STATE
7. What natural cycles does wildfire have an impact on?
HIGHT ENOUGH ENERGY THEY EMIT PROTONS SOME ELEMENTS REQUIRED HIGHER ENERGY
8. List at least three impacts of wildfire in San Diego.
WATER THAT IS WASTED, ANIMALS DYING, NATURE IS BURNING

This was a picture my partner and I took of our lab test with the strontium chloride. I think this is a really interesting picture because it shows how red the flame was. It also shows some of a our materials in this lab so the audience can get an idea of what equipment we used.
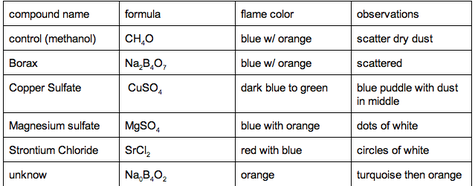
This was the table that me and my partner filled out after each experiment. It just is explaining what chemical we used,the formula,the color the flame turned once we lit it on fire and the observations we observed through the experiment.
Through out this lab I think it was a very interesting way to learn about chemicals and why flames turn different colors. I think it's better to do a hands on experiment then to just watch a video or look at pictures and read a book about it. I think the purpose was fulfilled through out this lab because like I said before it was hands on so we got to actually visualize what was going on. The experiment did work based on other groups. As in we all got about the same color flames.
My partner and I had about 2 errors and one of them was that the first experiment we did we both forget to use the stir stick to mix the methanal and the chemical and after lightning it on fire we observed our mistake so instead of redoing the experiment we just watched another groups and looked for a difference in flame color or different observations. Which there was no difference but after the first trial we adjusted it and made sure we didn't make the same mistake.
1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
Because the flame is a heat reaction so when you add a chemical and heat then theres a colored light but if you just have a chemical with out a reaction like fire then there won't be any type of reaction so in order to get the light which is a reaction you need something to cause that reaction, which is where the match comes in.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
when a metal ion and a nonmetal ion are combined they create an ionic bond.
3. What is the definition of a salt in chemistry?
Ionic compounds that can be formed by replacing one or more of the hydrogen ions of an acid with another positive ion.
4. What is fire?
a chemical reaction in which creates heat,light and smoke
5. What are the chemicals involved in lighting a match?
matches have a tip containing an oxidizer, potassium or sodium chlorate, and a fuel,
6. What is a chemical reaction?
A chemical reactions is when there is a cause and an effect. So if you have borax laying out and you add a flame there's ganna be a reaction because you added something to the chemical which will then create a reaction.
7. What natural cycles does wildfire have an impact on?
it affects the plant cycle,the life cycles of animals,water cycle. Plants burn which can no longer create photosythesis. Certain Animals or insects die which then affect other animals because thats what they conserve. Water Cycle is affected because the water that we can possibly use for drinking water is then used to put out the fires.
8. List at least three impacts of wildfire in San Diego.
water,animals,plants
My partner and I had about 2 errors and one of them was that the first experiment we did we both forget to use the stir stick to mix the methanal and the chemical and after lightning it on fire we observed our mistake so instead of redoing the experiment we just watched another groups and looked for a difference in flame color or different observations. Which there was no difference but after the first trial we adjusted it and made sure we didn't make the same mistake.
1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
Because the flame is a heat reaction so when you add a chemical and heat then theres a colored light but if you just have a chemical with out a reaction like fire then there won't be any type of reaction so in order to get the light which is a reaction you need something to cause that reaction, which is where the match comes in.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
when a metal ion and a nonmetal ion are combined they create an ionic bond.
3. What is the definition of a salt in chemistry?
Ionic compounds that can be formed by replacing one or more of the hydrogen ions of an acid with another positive ion.
4. What is fire?
a chemical reaction in which creates heat,light and smoke
5. What are the chemicals involved in lighting a match?
matches have a tip containing an oxidizer, potassium or sodium chlorate, and a fuel,
6. What is a chemical reaction?
A chemical reactions is when there is a cause and an effect. So if you have borax laying out and you add a flame there's ganna be a reaction because you added something to the chemical which will then create a reaction.
7. What natural cycles does wildfire have an impact on?
it affects the plant cycle,the life cycles of animals,water cycle. Plants burn which can no longer create photosythesis. Certain Animals or insects die which then affect other animals because thats what they conserve. Water Cycle is affected because the water that we can possibly use for drinking water is then used to put out the fires.
8. List at least three impacts of wildfire in San Diego.
water,animals,plants
